Abstract
Objective
Niclosamide is known to have anti-cancer and anti-inflammatory activities; however, its therapeutic mechanism has not been defined. In this study, to explain the therapeutic mechanism of niclosamide, we examined the effect of niclosamide on endothelial cell activation, leukocyte integration, proliferation, migration and angiogenesis in vitro.
Methods
Endothelia-leukocyte adhesion assays were used to assess primary cultures of human umbilical vein endothelial cells’ (HUVECs) activation following TNF-α treatment. Each step of angiogenesis was evaluated in vitro, including endothelial cell proliferation, migration and tube formation. Proliferation was examined using EdU assays, while wound migration assays and transwell assays were used to evaluate cell migration; cord like structure formation assays on Matrigel were used to assess tube formation. In vivo matrigel plug assay was used to assess angiogenesis. The protein expression was measured using western blot.
Results
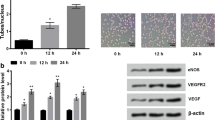
Niclosamide reduced the adhesion of human monocyte cells to HUVECs. Niclosamide also reduced protein expression of VCAM-1 and ICAM1 in HUVECs. Niclosamide significantly inhibited HUVEC proliferation, migration and cord-like structure formation. Niclosamide also suppresses VEGF-induced angiogenesis in vivo. Niclosamide attenuated IKK-mediated activation of NF-κB pathway in TNFα-induced endothelial cells. Niclosamide also suppresses VEGF-induced endothelial VEGFR2 activation and downstream P-AKT, P-mTOR and P-p70S6K.
Conclusions
Niclosamide exerted a potent effect on HUVECs activation, suggesting that it might function via an endothelia-based mechanism in the treatment of various diseases, including rheumatoid arthritis and cancer.








Similar content being viewed by others
References
Cines DB, Pollak ES, Buck CA, Loscalzo J, Zimmerman GA, McEver RP, et al. Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood. 1998;91:3527–61.
Pober JS, Sessa WC. Evolving functions of endothelial cells in inflammation. Nat Rev Immunol. 2007;7:803–15.
Risau W. Mechanisms of angiogenesis. Nature. 1997;386:671–4.
Costa C, Incio J, Soares R. Angiogenesis and chronic inflammation: cause or consequence? Angiogenesis. 2007;10:149–66.
Ono M. Molecular links between tumor angiogenesis and inflammation: inflammatory stimuli of macrophages and cancer cells as targets for therapeutic strategy. Cancer Sci. 2008;99:1501–6.
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420:860–7.
Rajashekhar G, Willuweit A, Patterson CE, Sun P, Hilbig A, Breier G, et al. Continuous endothelial cell activation increases angiogenesis: evidence for the direct role of endothelium linking angiogenesis and inflammation. J Vasc Res. 2006;43:193–204.
Gupta MK, Qin RY. Mechanism and its regulation of tumor-induced angiogenesis. World J Gastroenterol. 2003;9:1144–55.
Kalebic T, Garbisa S, Glaser B, Liotta LA. Basement membrane collagen: degradation by migrating endothelial cells. Science. 1983;221:281–3.
Bellon G, Martiny L, Robinet A. Matrix metalloproteinases and matrikines in angiogenesis. Crit Rev Oncol Hematol. 2004;49:203–20.
Tian Y, Jain S, Kelemen SE, Autieri MV. AIF-1 expression regulates endothelial cell activation, signal transduction, and vasculogenesis. Am J Physiol Cell Physiol. 2009;296:C256–66.
Ditzel J, Schwartz M. Worm cure without tears. The effect of niclosamide on taeniasis saginata in man. Acta Med Scand. 1967;182:663–4.
Osada T, Chen M, Yang XY, Spasojevic I, Vandeusen JB, Hsu D, et al. Antihelminth compound niclosamide downregulates Wnt signaling and elicits antitumor responses in tumors with activating APC mutations. Cancer Res. 2011;71:4172–82.
Jin Y, Lu Z, Ding K, Li J, Du X, Chen C, et al. Antineoplastic mechanisms of niclosamide in acute myelogenous leukemia stem cells: inactivation of the NF-kappaB pathway and generation of reactive oxygen species. Cancer Res. 2010;70:2516–27.
Wu CS, Li YR, Chen JJ, Chen YC, Chu CL, Pan IH, et al. Antihelminthic niclosamide modulates dendritic cells activation and function. Cell Immunol. 2014;288:15–23.
Liang L, Huang M, Xiao Y, Zen S, Lao M, Zou Y, et al. Inhibitory effects of niclosamide on inflammation and migration of fibroblast-like synoviocytes from patients with rheumatoid arthritis. Inflamm Res. 2015;64:225–33.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74.
Asahara T, Bauters C, Zheng LP, Takeshita S, Bunting S, Ferrara N, et al. Synergistic effect of vascular endothelial growth factor and basic fibroblast growth factor on angiogenesis in vivo. Circulation. 1995;92(II):365–71.
Niki T, Iba S, Tokunou M, Yamada T, Matsuno Y, Hirohashi S. Expression of vascular endothelial growth factors A, B, C, and D and their relationships to lymph node status in lung adenocarcinoma. Clin Cancer Res. 2000;6:2431–9.
Ivy SP, Wick JY, Kaufman BM. An overview of small-molecule inhibitors of VEGFR signaling. Nat Rev Clin Oncol. 2009;6:569–79.
Gupta SC, Sung B, Prasad S, Webb LJ, Aggarwal BB. Cancer drug discovery by repurposing: teaching new tricks to old dogs. Trends Pharmacol Sci. 2013;34:508–17.
Ley K, Laudanna C, Cybulsky MI, Nourshargh S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol. 2007;7:678–89.
Pohlman TH, Stanness KA, Beatty PG, Ochs HD, Harlan JM. An endothelial cell surface factor(s) induced in vitro by lipopolysaccharide, interleukin 1, and tumor necrosis factor-alpha increases neutrophil adherence by a CDw18-dependent mechanism. J Immunol. 1986;136:4548–53.
Pober JS, Sessa WC. Evolving functions of endothelial cells in inflammation. Nat Rev Immunol. 2007;7:803–15.
Kevil CG, Orr AW, Langston W, Mickett K, Murphy-Ullrich J, Patel RP, et al. Intercellular adhesion molecule-1 (ICAM-1) regulates endothelial cell motility through a nitric oxide-dependent pathway. J Biol Chem. 2004;279:19230–8.
Hou J, Baichwal V, Cao Z. Regulatory elements and transcription factors controlling basal and cytokine-induced expression of the gene encoding intercellular adhesion molecule 1. Proc Natl Acad Sci USA. 1994;91:11641–5.
Manning AM, Bell FP, Rosenbloom CL, Chosay JG, Simmons CA, Northrup JL, et al. NF-kappa B is activated during acute inflammation in vivo in association with elevated endothelial cell adhesion molecule gene expression and leukocyte recruitment. J Inflamm. 1995;45:283–96.
Sun X, Icli B, Wara AK, Belkin N, He S, Kobzik L, et al. MicroRNA-181b regulates NF-kappaB-mediated vascular inflammation. J Clin Invest. 2012;122:1973–90.
King ML, Lindberg ME, Stodden GR, Okuda H, Ebers SD, Johnson A, et al. WNT7A/Beta-catenin signaling induces FGF1 and influences sensitivity to niclosamide in ovarian cancer. Oncogene. 2015;34(26):3452–62.
Wieland A, Trageser D, Gogolok S, Reinartz R, Hofer H, Keller M, et al. Anticancer effects of niclosamide in human glioblastoma. Clin Cancer Res. 2013;19:4124–36.
Sack U, Walther W, Scudiero D, Selby M, Kobelt D, Lemm M, et al. Novel effect of antihelminthic Niclosamide on S100A4-mediated metastatic progression in colon cancer. J Natl Cancer Inst. 2011;103:1018–36.
Ferrara N. VEGF and the quest for tumour angiogenesis factors. Nat Rev Cancer. 2002;2:795–803.
Ivy SP, Wick JY, Kaufman BM. An overview of small-molecule inhibitors of VEGFR signaling. Nat Rev Clin Oncol. 2009;6:569–79.
Xia Y, Song X, Li D, Ye T, Xu Y, Lin H, et al. YLT192, a novel, orally active bioavailable inhibitor of VEGFR2 signaling with potent antiangiogenic activity and antitumor efficacy in preclinical models. Sci Rep. 2014;4:6031.
Matsuo M, Yamada S, Koizumi K, Sakurai H, Saiki I. Tumour-derived fibroblast growth factor-2 exerts lymphangiogenic effects through Akt/mTOR/p70S6kinase pathway in rat lymphatic endothelial cells. Eur J Cancer. 2007;43:1748–54.
Li W, Tan D, Zhang Z, Liang JJ, Brown RE. Activation of Akt-mTOR-p70S6 K pathway in angiogenesis in hepatocellular carcinoma. Oncol Rep. 2008;20:713–9.
Pang X, Yi Z, Zhang X, Sung B, Qu W, Lian X, et al. Acetyl-11-keto-beta-boswellic acid inhibits prostate tumor growth by suppressing vascular endothelial growth factor receptor 2-mediated angiogenesis. Cancer Res. 2009;69:5893–900.
Saraswati S, Kumar S, Alhaider AA. alpha-santalol inhibits the angiogenesis and growth of human prostate tumor growth by targeting vascular endothelial growth factor receptor 2-mediated AKT/mTOR/P70S6K signaling pathway. Mol Cancer. 2013;12:147.
Acknowledgments
The authors would like to thank Jinjin Fan for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest has been declared by authors.
Funding
This work is supported by grants from National Natural Science Foundation of China (Grant Number 81373182 and U1401222), Guangdong Natural Science Foundation (Grant Number S2011020002358, S2013010015363) and Guangdong Project of Science and Technology (Grant Number 2014A020212119).
Additional information
Responsible Editor: Bernhard Gibbs.
M. Huang and Q. Qiu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.




Rights and permissions
About this article
Cite this article
Huang, M., Qiu, Q., Zeng, S. et al. Niclosamide inhibits the inflammatory and angiogenic activation of human umbilical vein endothelial cells. Inflamm. Res. 64, 1023–1032 (2015). https://doi.org/10.1007/s00011-015-0888-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-015-0888-8