Abstract
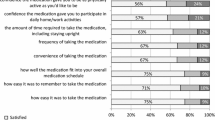
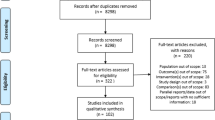
The purposes of the study were to review available published literature on magnitude of non-adherence with osteoporosis regimens and to determine the association between frequency and modality of medication administration with patient preference and adherence. We searched peer-reviewed journal databases—MEDLINE, EMBASE, Biosis and Derwent Drug File for publications (January 1979 to January 2009) including MeSH terms—“patient preference”, “adherence” and “compliance” based on “dosing frequency” and “modality”. Since adherence was difficult to accurately quantify, preference, compliance and persistence were evaluated. Patients’ preference and adherence at 12 months were higher with weekly over daily bisphosphonates (≥84% preference for weekly, medication possession ratios (MPR) 60–76% vs 46–64%; persistence 43.6–69.7% vs 31.7–55.7%). MPR reported for oral bisphosphonates were 68–71% at 12 months. At 2 years, only 43% of patients had MPR ≥80% for daily and weekly bisphosphonates. Observational studies (6–12 months) reported discontinuation rates of 18–22% for daily and 7% for weekly bisphosphonates. Data on monthly bisphosphonates are conflicting and confounded by cost differences, patient support programmes and definition of persistence. Studies suggest patient preference for annual zoledronic acid infusions over weekly bisphosphonates (66.4–78.8% vs 9.0–19.7%, respectively), but no data on compliance or persistence are available. Drug effectiveness, side effects and route of administration were more important than frequency. Although less frequent dosing is preferred, other factors such as perceived efficacy, side effects, medication cost, availability of patient support programmes and route of delivery are equally important. Adherence is complex and difficult to quantify and may not be exclusively influenced by frequency of medication administration.


Similar content being viewed by others
References
Lyles KW, Colon-Emeric CS, Magaziner JS et al (2007) Zoledronic acid and clinical fractures and mortality after hip fracture. N Engl J Med 357:1799–1809
World Health Organisation (2003) Adherence to long-term therapies. Evidence for action 2003. Available at www.who.int/chp/knowledge/publications/adherence_report/en/
Kinsella K, Velkoff V (2001) An aging world 2001. International population reports. Available at http://www.census.gov/prod/2001pubs/p95-01-1.pdf
Haynes RB, McDonald H, Garg AX, Montague P (2002) Interventions for helping patients to follow prescriptions for medications. Cochrane Database Syst Rev CD000011
Harrington JT, Ste-Marie LG, Brandi ML et al (2004) Risedronate rapidly reduces the risk for nonvertebral fractures in women with postmenopausal osteoporosis. Calcif Tissue Int 74:129–135
Harris ST, Watts NB, Genant HK et al (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. JAMA 282:1344–1352
Adami S, Felsenberg D, Christiansen C et al (2004) Efficacy and safety of ibandronate given by intravenous injection once every 3 months. Bone 34:881–889
Delmas PD, Recker RR, Chesnut CH 3rd et al (2004) Daily and intermittent oral ibandronate normalize bone turnover and provide significant reduction in vertebral fracture risk: results from the BONE study. Osteoporos Int 15:792–798
Reid IR, Brown JP, Burckhardt P et al (2002) Intravenous zoledronic acid in postmenopausal women with low bone mineral density. N Engl J Med 346:653–661
Black DM, Delmas PD, Eastell R et al (2007) Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med 356:1809–1822
Tucci JR, Tonino RP, Emkey RD, Peverly CA, Kher U, Santora AC 2nd (1996) Effect of three years of oral alendronate treatment in postmenopausal women with osteoporosis. Am J Med 101:488–501
Ettinger B, Black DM, Mitlak BH et al (1999) Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. Multiple Outcomes of Raloxifene Evaluation (MORE) Investigators. JAMA 282:637–645
O'Donnell S, Cranney A, Wells GA, Adachi JD, Reginster JY (2006) Strontium ranelate for preventing and treating postmenopausal osteoporosis. Cochrane Database Syst Rev CD005326
Inderjeeth CA, Foo AC, Lai MM, Glendenning P (2009) Efficacy and safety of pharmacological agents in managing osteoporosis in the old old: review of the evidence. Bone 44:744–751
Haynes RB (1979) Determinants of compliance: the disease and the mechanics of treatment. In: Haynes RB, Taylor DW, Sackett DL (eds) Compliance in health care. Johns Hopkins University Press, Baltimore, pp 49–62
Rand CS (1993) Measuring adherence with therapy for chronic diseases: implications for the treatment of heterozygous familial hypercholesterolemia. Am J Cardiol 72:68D–74D
Papaioannou A, Kennedy CC, Dolovich L, Lau E, Adachi JD (2007) Patient adherence to osteoporosis medications: problems, consequences and management strategies. Drugs Aging 24:37–55
Rabenda V, Mertens R, Fabri V et al (2008) Adherence to bisphosphonates therapy and hip fracture risk in osteoporotic women. Osteoporos Int 19:811–818
Compston J (2003) Do we need to monitor anti-osteoporosis treatement anyway? In: 30th European Symposium on Calcified Tissues. Rome, Italy
Reginster J, Minne HW, Sorensen OH et al (2000) Randomized trial of the effects of risedronate on vertebral fractures in women with established postmenopausal osteoporosis. Vertebral Efficacy with Risedronate Therapy (VERT) Study Group. Osteoporos Int 11:83–91
Black DM, Cummings SR, Karpf DB et al (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 348:1535–1541
Liberman UA, Weiss SR, Broll J et al (1995) Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. The Alendronate Phase III Osteoporosis Treatment Study Group. N Engl J Med 333:1437–1443
Ho AY, Kung AW (2005) Efficacy and tolerability of alendronate once weekly in Asian postmenopausal osteoporotic women. Ann Pharmacother 39:1428–1433
Ettinger B, Pressman A, Schein J, Chan J, Silver P, Connolly N (1998) Alendronate use among 814 women: prevalence of gastrointestinal complaints, non-compliance with patient instructions, and discontinuation. J Manag Care Pharm 4:488–492
Yood RA, Emani S, Reed JI, Lewis BE, Charpentier M, Lydick E (2003) Compliance with pharmacologic therapy for osteoporosis. Osteoporos Int 14:965–968
McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J (2004) Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas 48:271–287
Siris ES, Harris ST, Rosen CJ et al (2006) Adherence to bisphosphonate therapy and fracture rates in osteoporotic women: relationship to vertebral and nonvertebral fractures from 2 US claims databases. Mayo Clin Proc 81:1013–1022
Penning-van Beest FJ, Goettsch WG, Erkens JA (2004) Persistence with bisphosphonate therapy among post-menopausal osteoporotic women and the impact of dosing frequency. Value Health 7:724
Cowell W, Fulford-Smith A, Poultney S (2005) Adherence with bisphosphonate treatment for osteoporosis in UK patients. Bone 36(Suppl 2):S409–S410
Harris ST, Siris ES, Abbott TA, Barr CE, Rosen CJ (2005) Reduced osteoporotic fracture risk in patients adherence to bisphosphonate therapy. In: Program & Abstracts of The Endocrine Society’s 87th Annual Meeting; 2005 4–7 June, p 3–382
Reginster JY, Rabenda V (2006) Patient preference in the management of postmenopausal osteoporosis with bisphosphonates. Clin Interv Aging 1:415–423
International Osteoporosis Foundation (2005) IOF adherence report [online]. Available at http://www.osteofound.org/publications/pdf/adherence_gap_report.pdf
Claxton AJ, Cramer J, Pierce C (2001) A systematic review of the associations between dose regimens and medication compliance. Clin Ther 23:1296–1310
Richter A, Anton SE, Koch P, Dennett SL (2003) The impact of reducing dose frequency on health outcomes. Clin Ther 25:2307–2335, discussion 6
Simon JA, Lewiecki EM, Smith ME, Petruschke RA, Wang L, Palmisano JJ (2002) Patient preference for once-weekly alendronate 70 mg versus once-daily alendronate 10 mg: a multicenter, randomized, open-label, crossover study. Clin Ther 24:1871–1886
Kendler D, Kung AW, Fuleihan Gel H et al (2004) Patients with osteoporosis prefer once weekly to once daily dosing with alendronate. Maturitas 48:243–251
Baroutsou B, Babiolakis D, Stamatiadou AE et al (2004) Patient compliance and preference of alendronate once weekly administration in comparison with daily regimens for osteoporotic postmenopausal women. Ann Rheum Dis 63(Supple 1):455
Weiss M, Vered I, Foldes AJ, Cohen YC, Shamir-Elron Y, Ish-Shalom S (2005) Treatment preference and tolerability with alendronate once weekly over a 3-month period: an Israeli multi-center study. Aging Clin Exp Res 17:143–149
Cramer JA, Amonkar MM, Hebborn A, Altman R (2005) Compliance and persistence with bisphosphonate dosing regimens among women with postmenopausal osteoporosis. Curr Med Res Opin 21:1453–1460
Recker RR, Gallagher R, MacCosbe PE (2005) Effect of dosing frequency on bisphosphonate medication adherence in a large longitudinal cohort of women. Mayo Clin Proc 80:856–861
Sunyecz J, Gallagher R, MacCosbe PE (2006) Persistence with medication in women taking daily versus weekly bisphosphonates for osteoporosis. Female Patient 31:21–28
Ettinger MP, Gallagher R, MacCosbe PE (2006) Medication persistence with weekly versus daily doses of orally administered bisphosphonates. Endocr Pract 12:522–528
Brankin E, Walker M, Lynch N, Aspray T, Lis Y, Cowell W (2006) The impact of dosing frequency on compliance and persistence with bisphosphonates among postmenopausal women in the UK: evidence from three databases. Curr Med Res Opin 22:1249–1256
Silverman SL, Gold DT (2008) Compliance and persistence with osteoporosis therapies. Curr Rheumatol Rep 10:118–122
Weiss TW, McHorney CA (2007) Osteoporosis medication profile preference: results from the PREFER-US study. Health Expect 10:211–223
Weiss TW, Gold DT, Silverman SL, McHorney CA (2006) An evaluation of patient preferences for osteoporosis medication attributes: results from the PREFER-US study. Curr Med Res Opin 22:949–960
Duarte JW, Bolge SC, Sen SS (2007) An evaluation of patients’ preferences for osteoporosis medications and their attributes: the PREFER-International study. Clin Ther 29:488–503
Gold DT, Safi W, Trinh H (2006) Patient preference and adherence: comparative US studies between two bisphosphonates, weekly risedronate and monthly ibandronate. Curr Med Res Opin 22:2383–2391
Simon JA, Beusterien K, Hebborn A, Kline LN (2005) Bisphosphonate dosing preferences in women with postmenopausal osteoporosis: a study. Female Patient 30:31–36
Emkey R, Koltun W, Beusterien K et al (2005) Patient preference for once-monthly ibandronate versus once-weekly alendronate in a randomized, open-label, cross-over trial: the Boniva Alendronate Trial in Osteoporosis (BALTO). Curr Med Res Opin 21:1895–1903
Hadji P, Minne H, Pfeifer M et al (2008) Treatment preference for monthly oral ibandronate and weekly oral alendronate in women with postmenopausal osteoporosis: a randomized, crossover study (BALTO II). Joint Bone Spine 75:303–310
Weiss TW, Henderson SC, McHorney CA, Cramer JA (2007) Persistence across weekly and monthly bisphosphonates: analysis of US retail pharmacy prescription refills. Curr Med Res Opin 23:2193–2203
Silverman S, Cramer J, Sunyecz J et al (2007) Women are more persistent with monthly bisphosphonate therapy compared to weekly bisphosphonates: 12-month results from two retrospective databases [abstract W366]. In: 29th Annual Meeting of the American Society for Bone and Mineral Research; 2007 Sept 16–20; Honolulu, HI
Cooper A, Drake J, Drake J, Brankin E (2006) Treatment persistence with once-monthly ibandronate and patient support vs. once-weekly alendronate: results from the PERSIST study. Int J Clin Pract 60:896–905
Cooper A (2006) Compliance with treatment for osteoporosis. Lancet 368:1648
Richards JB, Cherkas LF, Spector TD (2007) An analysis of which anti-osteoporosis therapeutic regimen would improve compliance in a population of elderly adults. Curr Med Res Opin 23:293–299
Yood RA, Mazor KM, Andrade SE, Emani S, Chan W, Kahler KH (2008) Patient decision to initiate therapy for osteoporosis: the influence of knowledge and beliefs. J Gen Intern Med 23:1815–1821
McClung M, Recker R, Miller P et al (2007) Intravenous zoledronic acid 5 mg in the treatment of postmenopausal women with low bone density previously treated with alendronate. Bone 41:122–128
Saag K, Lindsay R, Kriegman A, Beamer E, Zhou W (2007) A single zoledronic acid infusion reduces bone resorption markers more rapidly than weekly oral alendronate in postmenopausal women with low bone mineral density. Bone 40:1238–1243
Fraenkel L, Gulanski B, Wittink D (2006) Patient treatment preferences for osteoporosis. Arthritis Rheum 55:729–735
Kendler DL, Bessette L, Hill CD et al (2009) Preference and satisfaction with a 6-month subcutaneous injection versus a weekly tablet for treatment of low bone mass. Osteoporos Int 21:837–846
Kothawala P, Badamgarav E, Ryu S, Miller RM, Halbert RJ (2007) Systematic review and meta-analysis of real-world adherence to drug therapy for osteoporosis. Mayo Clin Proc 82:1493–1501
Jackson RD, LaCroix AZ, Gass M et al (2006) Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med 354:669–683
Grant AM, Avenell A, Campbell MK et al (2005) Oral vitamin D3 and calcium for secondary prevention of low-trauma fractures in elderly people (Randomised Evaluation of Calcium Or vitamin D, RECORD): a randomised placebo-controlled trial. Lancet 365:1621–1628
Reginster JY, Rabenda V, Neuprez A (2006) Adherence, patient preference and dosing frequency: understanding the relationship. Bone 38:S2–S6
Inderjeeth CA, Glennon D, Petta A (2006) Study of osteoporosis awareness, investigation and treatment of patients discharged from a tertiary public teaching hospital. Intern Med J 36:547–551
Access Economics Pty Ltd (2010) A future less fragile. Available online at http://www.accesseconomics.com.au/publicationsreports
Conflicts of interest
This review was not sponsored. Dr. S Lee has no conflict of interest to declare. Dr. CA Inderjeeth has received funding not related to this project for research and/or educational grants from Merck Sharpe and Dohme, Sanofi Aventis and Servier, Speaker fee honoraria from Novartis, Sanofi Aventis and Servier and is on the advisory board for Amgen, Novartis and Sanofi Aventis. Dr. P Glendenning has received funding not related to this project for education grants and speaker fee honoraria from Eli Lilly, Merck Sharpe and Dohme, Novartis, Sanofi Aventis and Servier.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, S., Glendenning, P. & Inderjeeth, C.A. Efficacy, side effects and route of administration are more important than frequency of dosing of anti-osteoporosis treatments in determining patient adherence: a critical review of published articles from 1970 to 2009. Osteoporos Int 22, 741–753 (2011). https://doi.org/10.1007/s00198-010-1335-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-010-1335-x