Abstract
Summary
Efficacy of osteoporosis medication is not well-established among patients taking oral glucocorticoids. We assessed the efficacy of approved osteoporosis pharmacotherapies in preventing fracture by combining data from randomized controlled trials. Teriparatide, risedronate, and etidronate were associated with decreased vertebral fracture risk.
Introduction
Several osteoporosis drugs are approved for the prevention and treatment of glucocorticoid (GC)-induced osteoporosis. However, the efficacy of these treatments among oral GC users is still limited. We aimed to examine the comparative efficacy of osteoporosis treatments among oral GC users.
Methods
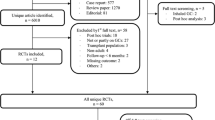
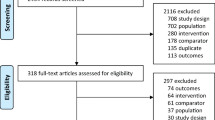
We updated a systematic review through to March 2015 to identify all double-blinded randomized controlled trials (RCTs) that examined osteoporosis treatment among oral GC users. We used a network meta-analysis with informative priors to derive comparative risk ratios (RRs) and 95 % credible intervals (95 % CrI) for vertebral and non-vertebral fracture and mean differences in lumbar spine (LS) and femoral neck (FN) bone mineral density (BMD). Treatment ranking was estimated using the surface under the cumulative ranking curve (SUCRA) statistic. A meta-regression was completed to assess a subgroup effect between patients with prior GC exposures and GC initiators.
Results
We identified 27 eligible RCTs examining nine active comparators. Etidronate (RR, 0.41; 95%CrI = 0.17–0.90), risedronate (RR = 0.30, 95%CrI = 0.14–0.61), and teriparatide (RR = 0.07, 95%CrI = 0.001–0.48) showed greater efficacy than placebo in preventing vertebral fractures; yet, no treatment effects were statistically significant in reducing non-vertebral fractures. Alendronate, risedronate, and etidronate increased LS BMD while alendronate and raloxifene increased FN BMD. In preventing vertebral fractures, teriparatide was ranked as the best treatment (SUCRA: 77 %), followed by risedronate (77 %) and zoledronic acid (76 %). For non-vertebral fractures, teriparatide also had the highest SUCRA (69 %), followed by risedronate (64 %). No subgroup effect was identified with regards to prior GC exposure.
Conclusions
Despite weak trial evidence available for fracture prevention among GC users, we identified several drugs that are likely to prevent osteoporotic fracture. Teriparatide, risedronate, and etidronate were associated with decreased vertebral fracture risk.


Similar content being viewed by others
Abbreviations
- BMD:
-
Bone mineral density
- CrI:
-
Credible interval
- GC:
-
Glucocorticoid
- LS:
-
Lumbar spine
- FN:
-
Femoral neck
- MCMC:
-
Markov Chain Monte Carlo
- RCT:
-
Randomized controlled trial
- SUCRA:
-
Surface Under the Cumulative RAnking curve
References
Overman RA, Yeh J-Y, Deal CL (2013) Prevalence of oral glucocorticoid usage in the United States: a general population perspective. Arthritis Care Res 65:294–298
Papaioannou A, Morin S, Cheung AM et al (2010) 2010 Clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. Can Med Assoc J 182:1864–1873
Freemantle N, Cooper C, Diez-Perez A et al (2013) Results of indirect and mixed treatment comparison of fracture efficacy for osteoporosis treatments: a meta-analysis. Osteoporos Int 24:209–217
Seibel MJ, Cooper MS, Zhou H (2013) Glucocorticoid-induced osteoporosis: mechanisms, management, and future perspectives. Lancet Diabetes Endocrinol 1:59–70
Grossman JM, Gordon R, Ranganath VK et al (2010) American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res 62:1515–1526
Van Staa TP, Laan RF, Barton IP et al (2003) Bone density threshold and other predictors of vertebral fracture in patients receiving oral glucocorticoid therapy. Arthritis Rheum 48:3224–3229
Hayashi K, Yamamoto M, Murakawa Y et al (2009) Bone fragility in male glucocorticoid-induced osteoporosis is not defined by bone mineral density. Osteoporos Int 20:1889–1894
de Nijs RNJ, Jacobs JWG, Lems WF et al (2006) Alendronate or alfacalcidol in glucocorticoid-induced osteoporosis. N Engl J Med 355:675–684
Adachi JD, Saag KG, Delmas PD et al (2001) Two-year effects of alendronate on bone mineral density and vertebral fracture in patients receiving glucocorticoids: a randomized, double-blind, placebo-controlled extension trial. Arthritis Rheum 44:202–211
Cohen S, Levy RM, Keller M et al (1999) Risedronate therapy prevents corticosteroid-induced bone loss: a twelve-month, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum 42:2309–2318
Roux C, Oriente P, Laan R et al (1998) Randomized trial of effect of cyclical etidronate in the prevention of corticosteroid-induced bone loss. Ciblos Study Group. J Clin Endocrinol Metab 83:1128–1133
Nawata H, Soen S, Takayanagi R et al (2005) Guidelines on the management and treatment of glucocorticoid-induced osteoporosis of the Japanese Society for Bone and Mineral Research (2004). J Bone Miner Metab 23:105–109
Gluer CC, Marin F, Ringe JD et al (2013) Comparative effects of teriparatide and risedronate in glucocorticoid-induced osteoporosis in men: 18-month results of the EuroGIOPs Trial. J Bone Miner Res 28:1355–1368
Cummings SR, Martin JS, McClung MR et al (2009) Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med 361:756–765
Kanis JA, Stevenson M, McCloskey EV, Davis S, Lloyd-Jones M (2007) Glucocorticoid-induced osteoporosis: a systematic review and cost-utility analysis. Health Technol Assess 11:1–231
Homik J, Cranney A, Shea B, et al. (2000) Bisphosphonates for steroid induced osteoporosis. Cochrane Database Syst Rev CD001347
Cranney A, Welch V, Adachi JD, et al. (2000) Calcitonin for the Treatment and Prevention of Corticosteroid-Induced Osteoporosis. Cochrane Database Syst Rev CD001983
Bucher H, Guyatt G, Griffith L, Walter S (1997) The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. J Clin Epidemiol 50:683–691
Caldwell DM, Ades AE, Higgins JPT (2005) Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ 331:897–900
Jadad AR, Moore RA, Carroll D et al (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17:1–12
Black D, Cummings S, Karpf D et al (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 348:1535–1541
Turner RM, Davey J, Clarke MJ, Thompson SG, Higgins JP (2012) Predicting the extent of heterogeneity in meta-analysis, using empirical data from the Cochrane Database of Systematic Reviews. Int J Epidemiol 41:818–827
Dias S, Sutton AJ, Ades AE, Welton NJ (2013) Evidence synthesis for decision making 2: a generalized linear modeling framework for pairwise and network meta-analysis of randomized controlled trials. Med Decis Making 33:607–617
Salanti G, Ades AE, Ioannidis JPA (2011) Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 64:163–171
Berger VW, Alperson SY (2009) A general framework for the evaluation of clinical trial quality. Rev Recent Clin Trials 4:79–88
Homik JE, Cranney A, Shea B et al (1999) A metaanalysis on the use of bisphosphonates in corticosteroid induced osteoporosis. J Rheumatol 26:1148–1157
Saag KG, Zanchetta JR, Devogelaer JP et al (2009) Effects of teriparatide versus alendronate for treating glucocorticoid-induced osteoporosis: thirty-six-month results of a randomized, double-blind, controlled trial. Arthritis Rheum 60:3346–3355
Ma YL, Zeng QQ, Chiang AY et al (2014) Effects of teriparatide on cortical histomorphometric variables in postmenopausal women with or without prior alendronate treatment. Bone 59:139–147
Abelson A, Ringe JD, Gold DT, Lange JL, Thomas T (2010) Longitudinal change in clinical fracture incidence after initiation of bisphosphonates. Osteoporos Int 21:1021–1029
Rosen C, Khosla S (2010) Placebo-controlled trials in osteoporosis—proceeding with caution. N Engl J Med 363:1365–1367
Acknowledgments
MAA is supported by a Canadian Network for Advanced Interdisciplinary Methods for Comparative Effectiveness Research (CAN-AIM) training scholarship and a Leslie Dan Faculty of Pharmacy Dean’s Entrance Scholarship, and was supported by a Drug Safety and Effectiveness Cross-Disciplinary Training scholarship. The funding agencies did not have any role in the design and conduct of the study; analysis or interpretation of the data; or preparation, review, or approval of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
JDA reports having participated in clinical trials sponsored by Procter & Gamble, Amgen and by Merck for the prevention and treatment of glucocorticoid-induced osteoporosis. JDA also reports having received consulting fees and research grants from Actavis, Amgen, Eli Lilly, Merck, and Novartis. MAA, JMA, MT, PP, LEL, and SMC state that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 1478 kb)
Rights and permissions
About this article
Cite this article
Amiche, M.A., Albaum, J.M., Tadrous, M. et al. Efficacy of osteoporosis pharmacotherapies in preventing fracture among oral glucocorticoid users: a network meta-analysis. Osteoporos Int 27, 1989–1998 (2016). https://doi.org/10.1007/s00198-015-3476-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-015-3476-4