Abstract
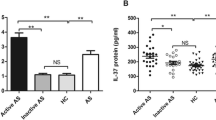
Interleukin-33 (IL-33) is implicated in rheumatoid arthritis with effects of promoting tumor necrosis factor-alpha (TNF-α) and interleukin-17 (IL-17) productions, which have been demonstrated to play a pivotal role in ankylosing spondylitis (AS). However, changes of IL-33 levels and its effects in AS have not been investigated. Eighty-nine and 178 healthy controls were included in the current study. Erythrocyte sedimentation rate, serum levels of C-reactive protein, IL-17, and IL-33 were determined. Effects of IL-33 on TNF-α and IL-6 productions were investigated. Effects of IL-33 on neutrophil migration were also evaluated. Serum levels of IL-33 were elevated in AS patients. Moreover, IL-33 was significantly higher in active AS patients according to Bath Ankylosing Spondylitis Disease Activity Index. IL-33 concentrations in serum were positively correlated with TNF-α and IL-17 levels (IL-33 and TNF-α, r = 0.54, P < 0.01; IL-33 and IL-17, r = 0.47, P < 0.01). IL-33 dose-dependently enhanced TNF-α and IL-6 productions by peripheral blood mononuclear cells (PBMCs) responding to lipopolysaccharide. IL-33 induced neutrophil migration only in higher doses (≥10 ng/ml). Serum levels of IL-33 were elevated in AS patients. IL-33 may play a role in AS development via enhancing TNF-α production by PBMCs and inducing neutrophil migration.




Similar content being viewed by others
References
Braun J, Sieper J (2007) Ankylosing spondylitis. Lancet 369:1379–1390
Mansour M, Cheema GS, Naguwa SM, Greenspan A, Borchers AT, Keen CL et al (2007) Ankylosing spondylitis: a contemporary perspective on diagnosis and treatment. Semin Arthritis Rheum 36:210–223
Wright C, Edelmann M, diGleria K, Kollnberger S, Kramer H, McGowan S et al (2009) Ankylosing spondylitis monocytes show upregulation of proteins involved in inflammation and the ubiquitin proteasome pathway. Ann Rheum Dis 68:1626–1632
Kim TH, Uhm WS, Inman RD (2005) Pathogenesis of ankylosing spondylitis and reactive arthritis. Curr Opin Rheumatol 17:400–405
Braun J, Bollow M, Neure L, Seipelt E, Seyrekbasan F, Herbst H et al (1995) Use of immunohistologic and in situ hybridization techniques in the examination of sacroiliac joint biopsy specimens from patients with ankylosing spondylitis. Arthritis Rheum 38:499–505
Toussirot E, Lafforgue P, Boucraut J, Despieds P, Schiano A, Bernard D et al (1994) Serum levels of interleukin 1-beta, tumor necrosis factor-alpha, soluble interleukin 2 receptor and soluble CD8 in seronegative spondylarthropathies. Rheumatol Int 13:175–180
Crew MD, Effros RB, Walford RL, Zeller E, Cheroutre H, Brahn E (1998) Transgenic mice expressing a truncated Peromyscus leucopus TNF-alpha gene manifest an arthritis resembling ankylosing spondylitis. J Interferon Cytokine Res 18:219–225
McLeod C, Bagust A, Boland A, Dagenais P, Dickson R, Dundar Y et al (2007) Adalimumab, etanercept and infliximab for the treatment of ankylosing spondylitis: a systematic review and economic evaluation. Health Technol Assess 11:1–158, iii–iv
Wendling D (2010) IL-23 and IL-17 in ankylosing spondylitis. Rheumatol Int 30:1547
Mei Y, Pan F, Gao J, Ge R, Duan Z, Zeng Z et al (2011) Increased serum IL-17 and IL-23 in the patient with ankylosing spondylitis. Clin Rheumatol 30:269–273
Kurowska-Stolarska M, Hueber A, Stolarski B, McInnes IB (2011) Interleukin-33: a novel mediator with a role in distinct disease pathologies. J Intern Med 269:29–35
Xu D, Jiang HR, Kewin P, Li Y, Mu R, Fraser AR et al (2008) IL-33 exacerbates antigen-induced arthritis by activating mast cells. Proc Natl Acad Sci 105:10913–10918
Matsuyama Y, Okazaki H, Tamemoto H, Kimura H, Kamata Y, Nagatani K et al (2010) Increased levels of interleukin 33 in sera and synovial fluid from patients with active rheumatoid arthritis. J Rheumatol 37:18–25
Matsuyama Y, Okazaki H, Hoshino M, Onishi S, Kamata Y, Nagatani K et al (2011) Sustained elevation of interleukin-33 in sera and synovial fluids from patients with rheumatoid arthritis non-responsive to anti-tumor necrosis factor: possible association with persistent IL-1β signaling and a poor clinical response. Rheumatol Int. doi:10.1007/s00296-011-1854-6
van der Linden S, Valkenburg HA, Cats A (1984) Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum 27:361–368
Wei JCC, Jan MS, Yu CT, Huang YC, Yang CC, Tsou HK et al (2007) Plasma homocysteine status in patients with ankylosing spondylitis. Clin Rheumatol 26:739–742
Hai H, Rudwaleit M, Braun J, Sieper J (2005) Six months open label trial of leflunomide in active ankylosing spondylitis. Ann Rheum Dis 64:124–126
Westergren A (1957) Diagnostic tests: the erythrocyte sedimentation rate range and limitations of the technique. Triangle 3:20–25
Boyum A (1964) Separation of white blood cells. Nature 204:793–794
Hsu YH, Li HH, Hsieh MY, Liu MF, Huang KY, Chin LS et al (2006) Function of interleukin-20 as a proinflammatory molecule in rheumatoid and experimental arthritis. Arthritis Rheum 54:2722–2733
Pease CT, Currey HL (1983) HLA-B27, ankylosing spondylitis and polymorph motility. Br J Rheumatol 22:172–176
Biasi D, Carletto A, Caramaschi P, Bellavite P, Andrioli G, Caraffi M et al (1995) Neutrophil functions, spondylarthropathies and HLA-B27: a study of 43 patients. Clin Exp Rheumatol 13:623–627
Guo L, Wei G, Zhu J, Liao W, Leonard WJ, Zhao K et al (2009) IL-1 family members and STAT activators induce cytokine production by Th2, Th17, and Th1 cells. Proc Natl Acad Sci 106:13463–13468
Carriere V, Roussel L, Ortega N, Lacorre DA, Americh L, Aguilar L et al (2007) IL-33, the IL-1-like cytokine ligand for ST2 receptor, is a chromatin-associated nuclear factor in vivo. Proc Natl Acad Sci 104:282–287
Schmitz J, Owyang A, Oldham E, Song Y, Murphy E, McClanahan TK et al (2005) IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 23:479–490
Hsu CL, Neilsen CV, Bryce PJ (2010) IL-33 is produced by mast cells and regulates IgE-dependent inflammation. PLoS One 5:e11944
Smith DE (2011) The biological paths of IL-1 family members IL-18 and IL-33. J Leukoc Biol 89:383–392
Nile CJ, Barksby E, Jitprasertwong P, Preshaw PM, Taylor JJ (2010) Expression and regulation of interleukin-33 in human monocytes. Immunology 130:172–180
Coremans IE, Daha MR, der Voort EAv, Siegert CE, Breedveld FC (1995) Subclass distribution of IgA and IgG antibodies against Clq in patients with rheumatic diseases. Scand J Immunol 41:391–397
Atagunduz P, Appel H, Kuon W, Wu P, Thiel A, Kloetzel PM et al (2005) HLA-B27-restricted CD8+ T cell response to cartilage-derived self peptides in ankylosing spondylitis. Arthritis Rheum 52:892–901
Verri WA Jr, Souto FO, Vieira SM, Almeida SC, Fukada SY, Xu D et al (2010) IL-33 induces neutrophil migration in rheumatoid arthritis and is a target of anti-TNF therapy. Ann Rheum Dis 69:1697–1703
Espinassous Q, Garcia-de-Paco E, Garcia-Verdugo I, Synguelakis M, von AS, Sallenave JM et al (2009) IL-33 enhances lipopolysaccharide-induced inflammatory cytokine production from mouse macrophages by regulating lipopolysaccharide receptor complex. J Immunol 183:1446–1455
Palmer G, Talabot-Ayer D, Lamacchia C, Toy D, Seemayer CA, Viatte S et al (2009) Inhibition of interleukin-33 signaling attenuates the severity of experimental arthritis. Arthritis Rheum 60:738–749
Disclosures
None.
Source of funding
This work was supported by Major Science and Technology Planning Project of Guangdong Province, China (No. 2003C30202).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Han, GW., Zeng, LW., Liang, CX. et al. Serum levels of IL-33 is increased in patients with ankylosing spondylitis. Clin Rheumatol 30, 1583–1588 (2011). https://doi.org/10.1007/s10067-011-1843-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-011-1843-x